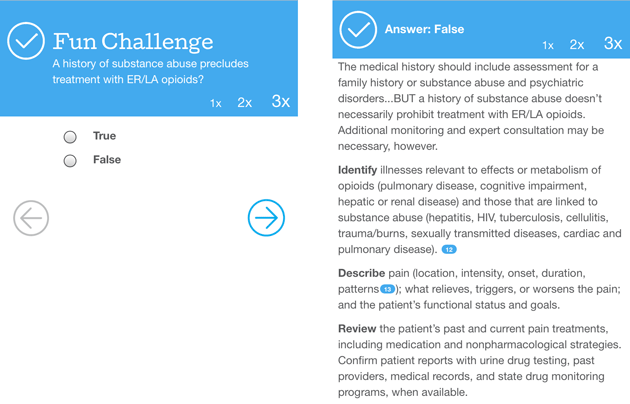
Mary Williams, a 42-year-old receptionist and mother of three, has a complicated medical record: She’s obese and diabetic, has a history of alcoholism, and smokes a pack a day. Substance abuse problems run in the family. She takes short-acting opioids every few hours for her lower back pain and neuropathy, but she’s still uncomfortable, so she goes back to the clinic. Should the doctor keep prescribing opioids?
Thousands of prescribers grapple with a variation of this question each day. Mary Williams is a made-up example in SCOPE of Pain, a continuing medical education class about the risks of prescribing opioids. As the death toll from overdoses soars, a growing number of states require prescribers to take such programs. SCOPE, a free module run by Boston University that can be completed online or in person, is a popular option: About 140,000 providers nationwide have taken it in the past five years.
Physicians who take SCOPE learn that Williams’ risk of opioid abuse is “moderate.” The course recommends increasing her dose of non-opioid painkillers and switching her to long-acting opioids that she should take every 12 hours. A year and a half later, participants learn, she’s thriving—going to pain support groups and not abusing the pills.
It sounds like a great resolution for a tricky case. The problem is, there’s no evidence that opioids work for chronic pain, according to guidelines released in 2016 by the Centers for Disease Control and Prevention. Indeed, a groundbreaking study in March found that opioids aren’t more effective for treating chronic pain than carefully calibrated cocktails of nonaddictive painkillers like Tylenol.
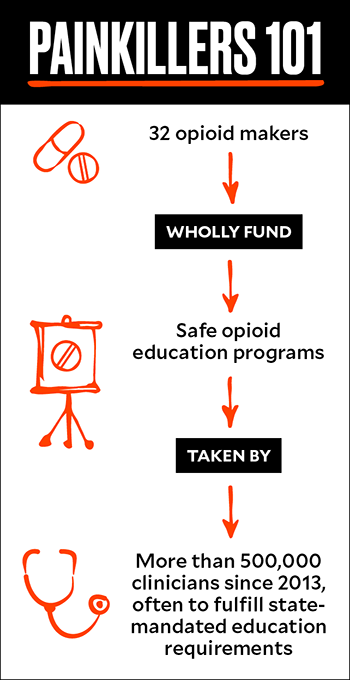
What might surprise the average clinician is that SCOPE, along with many of the most popular continuing medical education (CME) courses about opioids, is paid for wholly by opioid manufacturers, which are required by the Food and Drug Administration to fund such programs. A Mother Jones investigation found that the programs, including those affiliated with prestigious universities and professional organizations such as Johns Hopkins University and the American Society of Addiction Medicine, are backed by a conglomerate of extended-release opioid-makers, including Purdue Pharma, Pfizer, Endo, and Janssen. In some cases, such as Pri-Med’s SAFE Opioid Prescribing course, a partnership with the American College of Physicians, faculty have independently received hundreds of thousands of dollars from opioid manufacturers for independent consulting or research. The programs have a strikingly broad reach: More than half a million clinicians have participated since 2013.
Screenshots from Johns Hopkins’ app-based opioid education course, “Get SMART”
Within the so-called Risk Evaluation and Mitigation Strategies courses, there are common themes: The programs review overdose numbers and risks associated with specific brands. Some mention that 100 million Americans reportedly suffer from chronic pain—an Institute of Medicine number that has been heavily disputed. Some suggest practices that the CDC found little or no evidence supporting, such as tools that assess a patient’s risk of becoming addicted, or opioid rotation, a practice of switching patients from one opioid to another in order to avoid tolerance. According to the 2016 CDC guidelines, no studies have looked at the efficacy of this practice; evidence is “insufficient.”
Critics argue that the classes serve as an insidious form of pharmaceutical promotion. They give the impression that opioid manufacturers are taking responsibility for the epidemic—yet they also bear a chilling resemblance to the industry-funded education in the 1990s and 2000s that made opioid prescribing so popular to begin with.
For opioid-makers, CME “is one of the most important marketing [tactics] that they have,” says Dr. Adriane Fugh-Berman, a Georgetown pharmacology professor who studies industry influence on medicine. “It doesn’t look like advertising. It doesn’t look like promotion. It looks like education, and it’s required for most physicians.”
An FDA-mandated system
Twenty-eight states, from California to West Virginia, require that physicians or other prescribers take pain management or substance abuse CME, according to the National Alliance for Model State Drug Laws. That number is growing quickly—in 10 states, the rules have gone into effect since 2017. The courses play a key role in changing prescriber behavior. “There is very little time in the average clinician’s life to kick back and read a medical journal,” says Stanford psychiatry professor Dr. Anna Lembke. “They are too busy. They go to CME, and that’s where they get their information from experts.”
In practice, however, it’s hard to find pain management CME courses that fulfill these state requirements and aren’t funded by pharmaceutical companies. That’s because many common courses are Risk Evaluation and Mitigation Strategies (REMS)—classes that the FDA requires pharmaceutical companies to fund for select drugs (not just opioids) with “serious risks that may otherwise not be approved and available on the market.” The courses must be free or cheap, and they have to follow a basic blueprint curriculum outlined by the FDA. The makers of the powerful anti-acne drug Accutane, for example, are required to fund continuing education that warns the drug can cause birth defects.
But the REMS for opioids, mandated in 2012, marked the first time the FDA required education for such a broad swath of drugs—long-acting opioids, which pack high doses into pills that are supposed to release slowly over several hours—with so many complicated risks. Rather than each creating separate courses, the drug manufacturers pool millions of dollars and provide grants to about half a dozen programs, including SCOPE and SAFE. Some are online, while some are in-person lectures delivered at conferences across the country.
Federal law doesn’t require physicians to take REMS courses, but the classes have become a natural stand-in for state-mandated pain management requirements, given that they’re low cost and easily accessible and often fit the amount of training time—usually between one and three hours—that states require.
The courses disclose they are funded by “an independent educational grant” from the “Opioid Analgesic REMS Program Companies,” but many prescribers aren’t familiar with REMS and thus miss the pharmaceutical connection, says the California Health Care Foundation’s Kelly Pfeifer. “When you take one of these courses, you are left with the impression that if you do all this work—pill counts, pain agreements, risk assessments—that you will not do harm with opioids,” she adds.
Although the REMS programs follow the FDA blueprint, the conglomerate of pharmaceutical companies choose which CME programs, curricula, and faculty to fund. (REMS course representatives maintain that the classes are not influenced by pharmaceutical funding.) The FDA doesn’t audit the courses but requires that independent auditors review at least 10 percent of the offerings to make sure they’re following the blueprint and free of pharmaceutical influence.
REMS are particularly appealing to local hospital systems and municipalities scrambling to respond to skyrocketing overdose rates. “The REMS training was really a very convenient and seemingly well-informed response,” said Matt Willis, a public health officer for the Marin County health department in California. “Only in retrospect do I see that it may have been perpetuating the problem.”
Faculty ties to opioid-makers
While the courses themselves are funded by opioid manufacturers, some faculty also have individual financial ties with the pharmaceutical companies. Mother Jones reviewed federal data available through the Physician Payments Sunshine Act for the two dozen faculty listed as teachers for the FDA-mandated courses. Seven had received a combined $1.6 million between 2013 and 2016 from the opioid-makers: Drs. Charles Argoff, Jeffrey Gudin, Michael Brennan, Bill McCarberg, Steven Stanos, Oscar de Leon Casasola, and Lynn Webster. The funding paid for services like speeches, consulting, and research for a range of opioid and non-opioid medications.
Some of the faculty on the list have made news in recent years: Argoff and Stanos were cited in Sen. Claire McCaskill’s February investigation into pain advocacy groups for receiving roughly $600,000 and $90,000, respectively, from opioid manufacturers between 2013 and 2016. Brennan, of Fairfield, Connecticut, is among the top 10 Medicare prescribers of OxyContin in the country.
Faculty who responded to questions from Mother Jones maintain that their ties to pharmaceutical companies do not affect the content or teaching of the REMS courses. Many said they are simply following the FDA’s blueprint. “People who are sent to me have generally tried and failed any number of pain or curative treatments before seeing me,” said Brennan. He added that he encourages non-opioid therapies when appropriate, has more than 25 years of experience in treating pain, and has published numerous peer-reviewed articles on the topic. McCarberg said he no longer works for REMS courses; when he did, he, too, emphasized non-opioid pain treatments.
Webster, a pain doctor from Utah who contributed to SCOPE’s original content and developed the risk assessment tool that is recommended in many REMS courses, received more than $200,000 from opioid-makers between 2013 and 2016. He is named as a “key opinion leader” who helped drive the epidemic in a lawsuit recently filed by the state of Ohio against opioid manufacturers, as well as in a number of other state and district suits. “During a portion of his time as a KOL, Dr. Webster was under investigation for overprescribing by the U.S. Department of Justice’s Drug Enforcement Agency, which raided his clinic in 2010,” reads the Ohio complaint. “Although the investigation was closed without charges in 2014, more than 20 of Dr. Webster’s former patients at the Lifetree Clinic have died of opioid overdoses.”
Webster declined to comment on the lawsuit allegations. He noted that in the past, he has been retained by drug companies to perform research, but that he doesn’t benefit financially from any pharmaceutical products. “My interest is solely to advance new, and novel, therapies that benefit society,” he said. Dr. Dan Alford, the Boston University professor who directs SCOPE, said Webster “is a nationally recognized expert in safer opioid prescribing” whose pharmaceutical ties were disclosed when he consulted on SCOPE’s original curriculum in 2013. He added, “There’s as much as a firewall as there could be” between the course and its pharmaceutical funders. (Alford does not receive individual pharmaceutical funding, according to federal data.) When it comes to funding for CME, Alford pointed out, there aren’t many other options. “We would absolutely love it if we got funding from the federal government—if there was no pharmaceutical company involved in this at all.”
“Neurobiologically, they were built that way”
Of the seven faculty receiving the combined $1.6 million from opioid manufacturers that fund REMS courses, all but Webster are listed as faculty for SAFE, the collaboration between the American College of Physicians and Pri-Med that has been completed by 57,000 clinicians.
The course, which is free online, starts with Argoff, a neurologist at Albany Medical Center, talking about the rising number of opioid overdoses in a surprisingly defensive tone. “We need to keep in mind that illicit opioids—so not prescribed opioids only—significantly contributed to the increase,” he explains. “I’m urging all of us to have an open mind about how important it is to take care of our patients when opioid therapy is necessary…but not to directly link opioid therapy that’s prescribed for appropriate purposes to people dying from illicit drug use.” Yet health experts consistently say the opposite—and federal data shows that an estimated 4 out of 5 heroin users transitioned from prescription opioids. “Overprescription is responsible for the epidemic of opioids that we have,” Nora Volkow, director of the National Institute on Drug Abuse, said last year.
In another SAFE module, de Leon Casasola, a University of Buffalo anesthesiologist, infers that opioid addiction is caused by a missing neurological connection that occurs in some patients. “They cannot help it—neurobiologically, they were built that way,” he explains. Lembke, the Stanford professor, called the idea “pseudoscience.” De Leon Casasola did not respond to questions from Mother Jones.
The providers of SAFE, along with several other REMS courses, are audited by an umbrella CME group called the Accreditation Council for Continuing Medical Education (ACCME). Under FDA rules, ACCME is required to review at least 10 percent of SAFE courses. ACCME’s president, Dr. Graham McMahon, told me that pharmaceutical companies “have absolutely no say” in the courses, which are carefully reviewed to make sure that faculty aren’t covering topics that could be influenced by their financial ties. Yet when I brought up Argoff and de Leon Casasola’s statements, he declined to comment. Instead, he suggested that if there were components of courses that seemed concerning, I should submit them to ACCME’s complaints portal.
Some REMS courses are more strict when it comes to pharmaceutical influence: CORE, the largest opioid REMS course and a partnership between 11 health organizations, including the American Society of Addiction Medicine (ASAM) and Medscape, has a policy that prohibits faculty advisory committee members from having financial ties to opioid manufacturers. The course uses the FDA blueprint as a jumping-off point, says ASAM legislative chair Dr. Corey Waller, and has added sections about opioid addiction and non-pharmacological pain treatments. Still, critics like Lembke say that by virtue of being a REMS course, it’s inherently opioid-focused. A sample “knowledge test” question from CORE:

Screenshot from “Opioid Prescribing: Safe Practice, Changing Lives,” a CORE course
(“It was a poorly designed question,” acknowledges Waller, adding that A and B are more appropriate answers.)
Ramping up REMS
Many REMS critics say the problem extends beyond the opioid manufacturers to the FDA’s blueprint, which was subject to years of negotiation. After the agency solicited public comment to an initial blueprint draft in 2011, trade groups, pharmaceutical companies, and individuals submitted dozens of public responses. Over time, the draft changed in subtle ways. An early version stated, “Prescribers should assess each patient’s risk of abuse, including history of substance use and serious mental illness.” The final version featured an addition that would pertain to the fictitious Mary Williams: “A history of substance abuse does not prohibit treatment with [extended-release] opioid analgesics but may require additional monitoring and expert consultation.”
University of Washington medical school professor Mark Sullivan struggled with the REMS requirements. In 2014, Sullivan received a REMS grant for more than $600,000. He already delivered a pain management course to university residents, who often came to him flummoxed by what to do about all the incoming chronic pain patients, and he figured the funding would help reach a broader audience. But he soon found that the course wasn’t particularly relevant to the learners. “They would ask, ‘Why is this all about opioids?’” he recalls. “The core sentiment, and this was not ever explicitly stated in the program, was that you can have safer opioid prescribing without having less opioid prescribing.” Sullivan and his team tried to incorporate information about tapering off opioids and using non-pharmacological options, but “that was not what we were being paid to do,” he told me. REMS stopped funding Sullivan’s program in 2015.
Spokesmen for several REMS courses argue there simply aren’t great alternatives to opioids. “Everything we use to treat chronic pain lacks adequate evidence, including all pharmacologic and non-pharmacologic approaches,” noted Alford, the SCOPE director. “Unfortunately, we cannot wait for robust evidence to become available before we start treating patients suffering from severe chronic pain.”
In the meantime, REMS is expanding. Late last year, FDA Commissioner Scott Gottlieb announced that REMS courses will soon be required not only for manufacturers of extended-release opioids, but for makers of immediate-release opioids as well—adding 277 medications to the current 64. The agency also released a new draft blueprint, to be finalized by the end of the year, that incorporates information about addiction and non-opioid pain treatment.
Once again, public comment has opened the floodgates. Industry group Academy of Integrative Pain Management objected “to the use of the inflammatory term ‘epidemic,’” preferring instead the “problem of prescription opioid drug abuse.” (The organization received more than $1 million from Purdue Pharma between 2012 and 2017, according to McCaskill’s report.) Meanwhile, nonprofit Physicians for Responsible Opioid Prescribing noted that the blueprint draft continues to omit the fact that there is no evidence supporting the treatment of chronic pain with opioids.
Recently, a small group of advocates including Lembke and Fugh-Berman have been calling for radical change in how pain education is structured: Rather than REMS, physicians should have the option to take government-funded courses that have no connection to pharmaceutical companies. Fugh-Berman has collaborated with peers at George Washington University to create such a program: the DC Center for Rational Prescribing, a slew of online CME courses funded by DC’s public health department. It’s still a small operation—taken by about 1,500 prescribers so far—but the courses have a distinctly different feel than REMS does, with modules including tapering, non-opioid alternatives to pain, medical cannabis, and even one titled “Industry Influence on the Practice of Medicine.”
Fugh-Berman says there are plenty of other ways in which pharmaceutical companies could help that wouldn’t serve as a potential marketing platform: put research funding in a blind trust controlled wholly by the National Institutes of Health, or fund proven addiction medications like buprenorphine or methadone.
“But would they do it with no chance of getting anything in return?” she said. “I don’t think they would.”
Note: This article has been updated.
















